Efficacy / Effect
1. Children:
1) Growth failure due to pituitary growth hormone secretion disorder
2) Growth failure due to Turner syndrome
3) Growth failure due to chronic renal failure
4) Improvement of growth and body composition in children with Prader-Willi syndrome
5) Failure to thrive in short children with small for gestational age (SGA)
6) Failure to thrive in pediatric patients with idiopathic short stature (ISS)
2. Adults: As an growth hormone replacement therapy for adults with growth hormone deficiency identified by the two dynamic test, the patient must meet the following criteria:
1) Childhood onset deficiency (Childhood Onset): Patients diagnosed with growth hormone deficiency in childhood must be re-evaluated and confirmed to be growth hormone deficiency before starting growth hormone replacement therapy.
2) Adult onset deficiency (Adult Onset): Before starting growth hormone replacement therapy, secondary growth hormone deficiency due to hypothalamic or pituitary disorder and at least one other hormone deficiency (excluding prolactin) should be diagnosed and appropriate replacement therapy should be in progress.
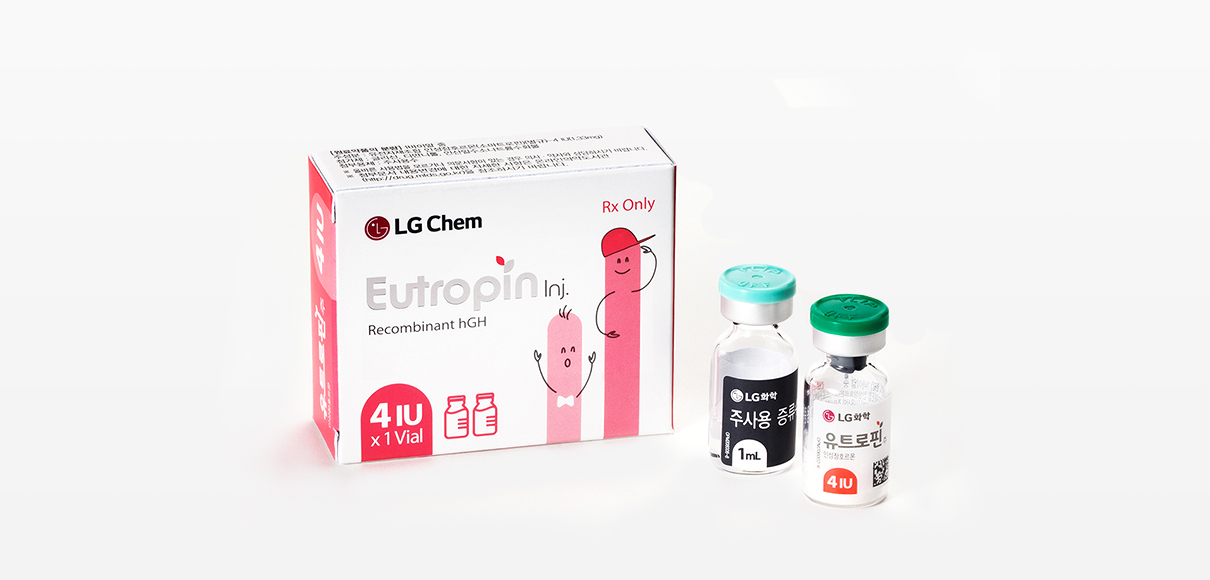
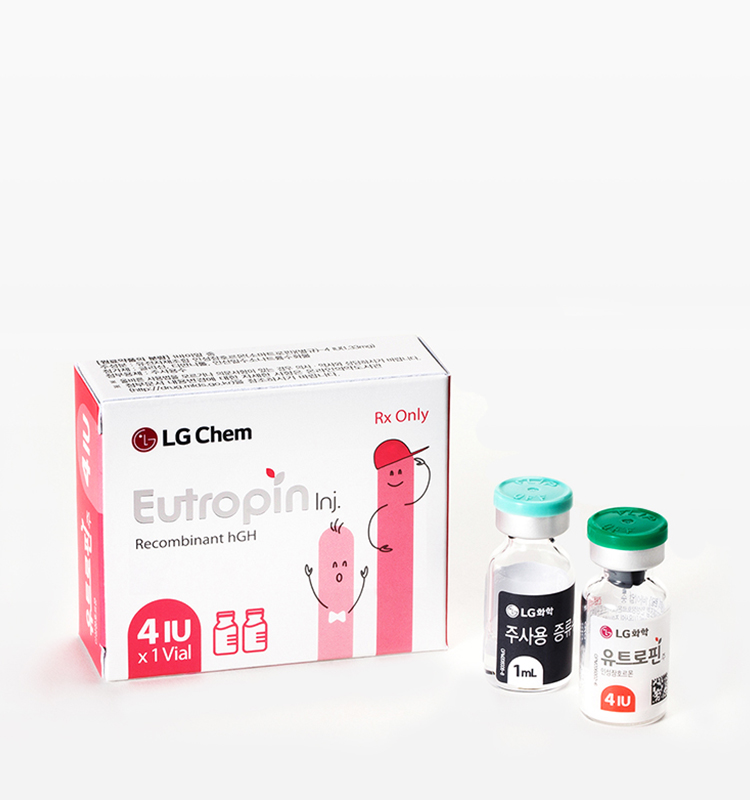
Active Ingredient and Capacity
Somatropin(recombinant human growth hormone) 4IU
etc.
Appearance & Packaging Unit
White or almost white lyophilized powder filled in a vial and a colorless and transparent liquid preparation when dissolved in accompanying solvent.
1V: [vial(4IU) x 1 pc. + accompanying solvent(1mL) x 1 pc.]/box
5V: [vial(4IU) x 5 pcs. + accompanying solvent(1mL) x 5 pcs.]/box
Storage
In a hermetic container, keep refrigerated (2~8℃).
Advanced Adhesive Products and Their Functions