Efficacy / Effect
For active primary immunization against diphtheria, tetanus, pertussis, hepatitis B and Haemophilus influenzae type b disease in infants from 6 weeks of age.
Usage/Dosage
Recommended Dosage: Intramuscular injections of 3 doses (1 dose=0.5 mL) at 6, 10, and 14 weeks of age
etc.
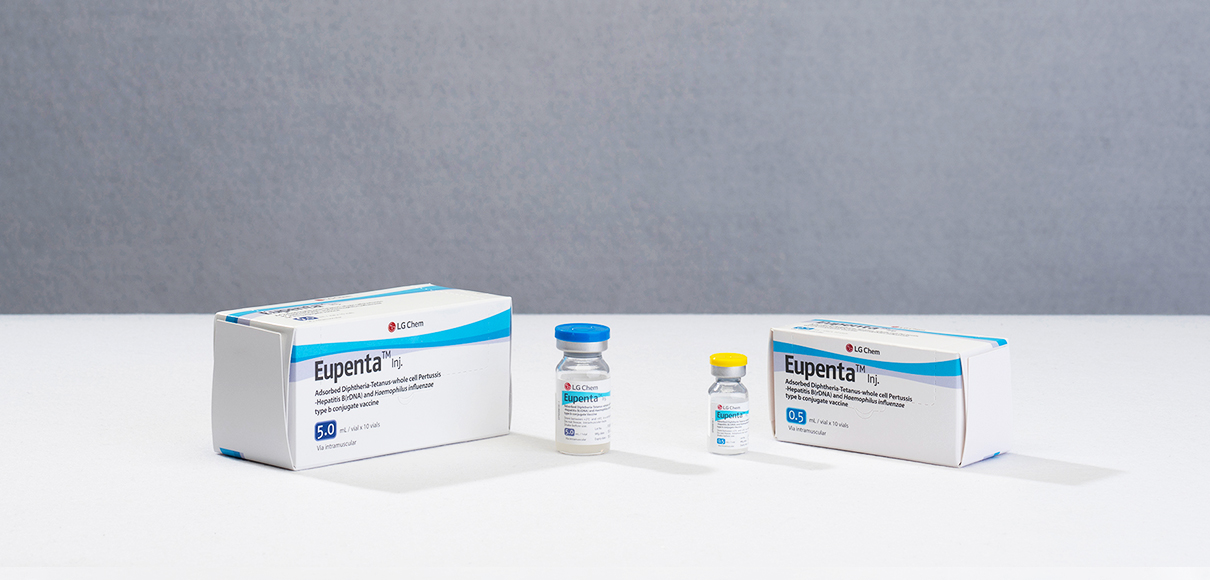
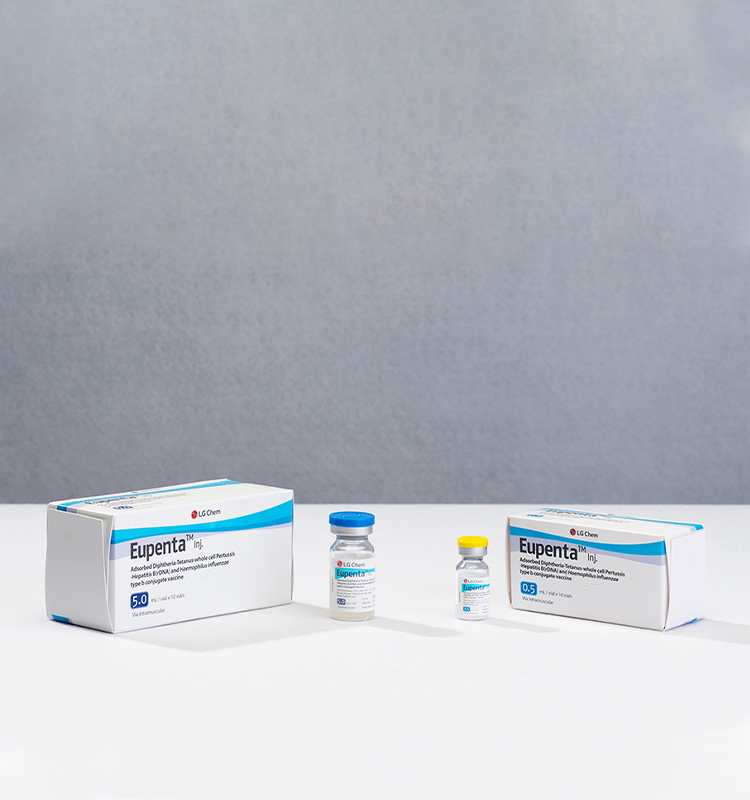
Appearance & Packaging Unit
Slightly opaque white suspension for injection
0.5 mL x 10 vials
5.0 mL x 10 vials
Active Ingredient and Capacity
Each 0.5 mL of EupentaTM Inj. contains 15 Lf of diphtheria toxoid, 10 Lf of tetanus toxoid, not less than 4 IU of inactivated pertussis suspension, 10 μg of purified hepatitis B surface antigen and 30~50 μg of purified capsular polysaccharide of Haemophilus influenzae type B conjugated to the tetanus toxoid.
Storage
Store at 2~8°C. Do not freeze.
Advanced Adhesive Products and Their Functions